J. A. Wertheim; H. Petrowsky; S. Saab; J. W. Kupiec-Weglinski; R. W. Busuttil
Authors and DisclosuresPosted: 10/03/2011; American Journal of Transplantation. 2011;11(9):1773–1784 © 2011 Blackwell Publishing
Abstract
Liver transplantation is the gold standard of care in patients with end-stage liver disease and those with tumors of hepatic origin in the setting of liver dysfunction. From 1988 to 2009, liver transplantation in the United States grew 3.7-fold from 1713 to 6320 transplants annually. The expansion of liver transplantation is chiefly driven by scientific breakthroughs that have extended patient and graft survival well beyond those expected 50 years ago. The success of liver transplantation is now its primary obstacle, as the pool of donor livers fails to keep pace with the growing number of patients added to the national liver transplant waiting list. This review focuses on three major challenges facing liver transplantation in the United States and discusses new areas of investigation that address each issue: (1) the need for an expanded number of useable donor organs, (2) the need for improved therapies to treat recurrent hepatitis C after transplantation and (3) the need for improved detection, risk stratification based upon tumor biology and molecular inhibitors to combat hepatocellular carcinoma.
Introduction
Experimental liver transplantation was first attempted in 1955 when Stuart Welch from Albany, New York, implanted an auxiliary liver into the paravertebral fossa of a dog. One year later Jack Cannon at the University of California, Los Angeles, performed the first orthotopic liver transplant, also in the canine species. These experimental milestones put in motion the intimate relationship between scientific investigation and clinical progress that began with Thomas Starzl's first human liver transplant in 1963.
[1] We have learned much from those pioneering efforts, and today liver transplantation is the established treatment for all types of end-stage liver disease, both acute and chronic, as well as primary liver cancer in the setting of cirrhosis.
This review focuses on current clinical and research initiatives aimed at addressing the greatest challenges in liver transplantation today. The annual number of recovered deceased donor livers has decreased nationwide from 7017 in 2006 to 6608 in 2010, according to data collected by the Organ Procurement and Transplantation Network (OPTN).
This trend mirrors the annual liver transplantation rate and reinforces the fact that liver transplantation in the United States is driven, and limited, by the supply of usable donor organs. Part 1 of this review addresses the organ shortage dilemma and presents current and novel strategies to expand the useable organ pool.
As hepatitis C (HCV) is the most common indication for liver transplant, part 2 of this review discusses HCV recurrence in the posttransplant setting. HCV is a common cause for graft failure that is related to the difficulty in treating patients with recurrent disease due to decompensated liver function and debilitating side effects of anti-HCV medications. New protease inhibitors are on the horizon, and are discussed within this report. The past 10 years have also witnessed an increase in liver transplantation for hepatocellular carcinoma (HCC).
Part 3 addresses the need for improved molecular analysis of HCC to develop improved risk stratification methods and molecular inhibitors. Proper patient selection is critical to posttransplant survival; however, today selection is based primarily on tumor size and number which does not always accurately predict tumor biology. To that end, we identify the development of new biomarkers as a critical challenge that may one day serve as a personalized molecular signature to more accurately select HCC candidates for liver transplantation and provide an optimal treatment regimen of molecular inhibitors to combat advanced HCC.
Lack of Quality Donor Organs
One of the greatest hurdles that liver transplantation faces today is the growing discrepancy between the demand for, and availability of, donor livers. Although this gap has been slightly reduced since the introduction of the Model for end-stage liver disease (MELD) allocation system in the United States in 2002, there remains twice as many patients on the waiting list in 2008 (12 230 patients) compared to those who were transplanted that same year (6,319 recipients) (Figure 1).
Figure 1
Gap between supply and demand for useable donor livers. Annual number of listed liver transplant candidates at the end of the year (solid circle) are compared to the total liver transplantations (open triangle) performed in the United States since the introduction of the MELD allocation system. Data were retrieved from the 2009 OPTN/SRTR Annual Report (94).
Different strategies have evolved to expand the donor pool (
Table 1).
Table 1.
Strategies and techniques to expand the donor organ pool
Use of donor livers with extended criteria
Use of steatotic donor organs
HCV-positive donor organs for HCV-positive recipients
Use of high-risk CDC donor organs
Donation after cardiac death
Split liver transplantation
Living donor liver transplantation
Domino liver transplantation
Marginal grafts or extended criteria donor (ECD) organs from brain dead donors represent the largest potential for donor pool expansion today. Although liver transplantation in the United States is driven by the supply of whole organs from donation after brain death (DBD), approximately 12% of all liver transplant recipients in 2008 received partial grafts from living donors, split grafts from DBD, or whole organs from donation after cardiac death (DCD) (Figure 2).
Figure 2.
Distribution of liver transplantations performed in the United States in 2008 according to type of donor organ. DBD = donation after brain death; DCD = donation after cardiac death; LDLT, living donor liver transplantation. Data were retrieved from the 2009 OPTN/SRTR Annual Report (94).

Despite efforts to expand this resource, the total annual number of organs recovered from this pool remained constant at 700–800 per year over the past 4 years (Figure 3).
Figure 3.
Distribution of alternative liver grafts (donation after cardiac death, living donor split grafts and split grafts from brain dead donors) between 1999 and 2008. Annual number of liver transplantations performed with partial grafts from living donors (open triangle), split grafts from brain death donors (open circle), and full grafts from donation after cardiac death (solid circle ). The total number of this pool is represented by theopen square. Data were retrieved from the 2009 OPTN/SRTR Annual Report (94).
Challenge of ECD Organs:
Although UNOS has defined the standard for ECD kidneys, a universal definition for livers has not yet been established. However, advanced donor age, steatosis and prolonged hospital stay are the most important criteria defining ECD organs by many centers. Donor age,[3–6] cold ischemia time[3,4] and donor hospital stay[3] are significant donor variables in models predicting outcome after liver transplantation. Although each criterion is not a contraindication for transplantation, the accumulation of several criteria significantly increases the risk for poor outcome after liver transplantation.[3,5] One drawback of the prediction models is that none include parenchyma quality such as steatosis. Steatosis, especially macrosteatosis, is an established risk factor for primary nonfunction and graft dysfunction. Microsteatosis, which often appears as a mixed form with macrosteatosis, has a smaller effect on ischemia-reperfusion injury than macrostatosis.[7] While mild macrosteatosis (less then 30%) is acceptable for use in transplantation if cold ischemia time is short, the allocation of livers with moderate macrosteatosis (30–60%) remains challenging[6] due to the significantly increased risk of primary graft failure.
A national analysis of the Scientific Registry of Transplant Recipients revealed that macrosteatosis more then 30% is an independent risk factor for reduced 1-year graft survival.
[8] Additionally, the epidemics of obesity and diabetes in the United States during the past two decades have led to increased donor livers with steatosis.
[9,10] However, the utilization of steatotic livers can significantly contribute to the expansion of the donor pool. Although many predictive donor variables cannot be changed (donor age, hospital stay, etc.) once an offer is made, minimizing cold ischemia and appropriate donor-recipient matching should be a primary consideration when dealing with ECD or steatotic organs for transplantation. Pharmacological protection
[11] and machine perfusion of the liver
[12–14] are promising protective strategies against ischemia-reperfusion injury that are particularly applicable to high risk organs. These strategies attempt to rehabilitate marginal grafts in an attempt to expand the donor pool.
Challenge of Living Donor Liver Transplantation: Living donor liver transplantation (LDLT) peaked in 2001 in the United States with 524 cases (Figure 3).
The following year, 363 living donor liver transplants were performed, and the living donation rate declined annually until 2010 when the rate increased almost 30% above 2009 levels (OPTN data). In the United States, approximately 20% of these organs are given to pediatric recipients. LDLT is a life saving procedure especially for pediatric patients and adult patients who are otherwise disadvantaged by the current MELD allocation system. However, the risk to a healthy donor is not zero as demonstrated by the recent deaths of two living donors in the United States in 2010. Perioperative mortality in LDLT is estimated at 0.15–0.30% and may be slightly higher (0.5%) for right hemi-liver donation.
[15] In addition, recent reports demonstrate that LDLT is associated with significant postoperative complications in both living donors and their recipients.
[16,17]
The multicenter Adult-to-Adult Living Donor Liver Transplantation cohort Study (A2ALL)
[16] and a report from the University of Pittsburgh
[17] demonstrated that living donors experienced severe or life-threatening complications in 35–12% of these cases, respectively. In terms of recipient morbidity, biliary complications (32% vs. 10%), unplanned reexploration (26% vs. 17%), hepatic artery thrombosis (6.5% vs. 2.3%) and portal vein thrombosis (2.9% vs. 0%) occurred more frequently in recipients from living compared to deceased donors.
[18] A recently published study from the University of Pittsburgh revealed a higher biliary (42%) and vascular (15%) complication rate in LDLT recipients.
[17] The possibility of donor death, the higher rate of perioperative complications and the safer alternative of deceased donor liver transplantation most likely explain why LDLT declined in the United States over the past several years (Figure 3). Before further extending the living donor pool, the transplantation societies and the transplant community have to define how much risk we are willing to accept in LDLT for both donor and recipient.
[19]Challenge of Split Liver Transplantation:
Split liver transplantation is an established method to extend the donor pool by creating two partial grafts from one donor organ which can be given to two recipients. This complex procedure requires detailed knowledge of liver anatomy and expertise in hepatobiliary surgery. The most widely used splitting technique in the United States is the division of the liver into a right trisectoral graft (segments 1, 4–8) for an adult recipient and a left lateral sectoral graft (segments 2 and 3) for a pediatric recipient.
The introduction of this technique has dramatically decreased the need for living donation at many pediatric transplant centers.[20] Our results with over 250 split liver transplants illustrate that proper donor selection with appropriate parenchymal volume can yield equivalent survival compared to whole organ transplantation.[21] Moreover, biliary and arterial complications are not significantly different in segmental grafts compared to whole organs.[22,23] Despite the shortfall between deceased donor whole organ availability and recipient demand, split liver transplantation remains at only 4% to 5% of all livers used for transplantation (Figure 2 and 3).
A survey in the United States on split liver transplantation showed thatex vivo splitting was performed in 54% and in situin 46% with equivalent outcome.
[24] However, ex vivo splitting when performed in the recipient's institution appears inferior in terms of cold ischemia time if the graft is shared between two centers. Further limiting factors and future challenges are the learning curve, logistical demands and restrictive organ allocation policies in some regions. However, split liver transplantation of deceased donor organs has a high-growth potential if optimal donors can be identified and procurement teams can be assembled with the necessary expertise to effectively split the organ.
Challenge of DCD Organs:
Persistent organ shortage and increasing deaths on the waiting list have resulted in an increased utilization of livers from controlled DCD in the United States over the past 10 years (Figure 3). During 2005 to 2008, DCD liver transplantation reached a plateau and remained at 4–5% of all liver transplants. It is well documented that DCD liver transplantation has an inferior outcome compared to DBD liver transplantation in terms of biliary complications, graft survival and need for retransplantation.
[25–27] Although overall patient survival appears similar between DCD and DBD liver transplantation, 1-year graft survival was significantly reduced for DCD allografts (60 vs. 82%), furthermore retransplantation was required in 15% of the cases within the first year of transplantation.
[26] Indications for retransplantation were primary nonfunction, delayed graft failure, ischemic-type biliary strictures and hepatic artery thrombosis. It is conceivable that this inferior outcome translates into higher costs. The largest single-center study demonstrated that the total cost per patient was 20% higher for DCD compared to DBD liver transplantation.
[26]
A recently published national study identified donor (age more then 50 years, weight more then 100 kg, donor warm ischemia time more then 35 min) and recipient (age more then 55 years, male gender, African American race, HCV positivity, metabolic disorder, MELD more then 35, hospitalization at transplantation and mechanical treatment) risk factors predictive of graft failure.
[28] These risk factors demonstrate that careful donor-recipient matching is of paramount importance for successful outcome of DCD liver transplantation.
Recent reports from the Cleveland Clinic suggest that the administration of tissue plasminogen activator into the donor hepatic artery before implantation might reduce the rate of ischemic-type biliary strictures.[29] As long as patients are dying on the waiting list, it is hard to ignore that DCD liver transplantation has saved many lives even in light of moderate inferior outcome and slightly higher costs. If the outcome of DCD liver transplantation can be improved to the level of DBD liver transplantation, DCD would probably have the greatest growth potential compared to split liver transplantation and LDLT. Therefore, every effort should be promoted in this field to achieve this life-saving goal.
Recurrence of HCV After Liver Transplantation
HCV is the most common indication for liver transplantation in the United States, accounting for approximately 40–45% of all transplants.[30] After liver transplantation, infection of the transplanted liver is universal and recurrent HCV directly impacts patient and graft survival.[31,32] Although short-term survival does not appear to be affected by recurrent infection, differences in survival become significant with long-term follow-up.[32–34]
Recurrent histological damage can be seen within 3 months of transplantation.
[35] Approximately 20–30% of patients progress to cirrhosis and graft failure and either require retransplantation or face a high likelihood of death within 5 years of transplant.
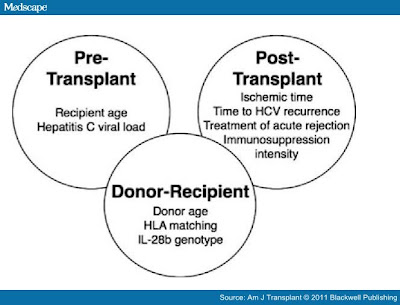
[34,36] Several factors appear to affect the severity and time to recurrent infection and are stratified into donor, recipient, operative and viral factors (Figure 4).
Pretransplant and posttransplant viral load affects HCV recurrence; viral loads above ~1×106 IU/mL around the time of transplantation are associated with a decreased 5-year survival. Approximately 57% of patients were alive at 5 years in the more then 1×106 IU/mL cohort versus 84% in the less then 1×106 IU/mL group.[37,38] Unfortunately, efforts to decrease viral load have been limited by concerns of precipitating hepatic decompensation.
Figure 4.
Factors associated with recurrent hepatitis C disease after liver transplantation.
Challenge of Balancing Graft Rejection and HCV Recurrence:
An important challenge in treating recipients transplanted for HCV is monitoring for disease progression as neither serum transaminases nor HCV RNA viral load correlate with clinical disease nor overall hepatic function.[39] Histological confirmation of HCV reinfection is the gold standard test for disease recurrence. Liver biopsies are associated with risk of pain and bleeding. Generally, antiviral treatment is initiated in patients with stage II or more severe fibrosis. As an alternative to biopsies, noninvasive markers of fibrosis such as Fibrospect II, Fibrosure or radiological studies such as Fibroscan and MRI elastrography are attractive considerations to monitor the degree of fibrosis.[40] However, these novel markers of fibrosis require further validation in the posttransplant setting.
There is increased understanding of the relationship between HCV infection and the immune system. Emerging evidence suggest that regulatory T cells may play an integral role. There is an enhanced expression of regulatory T cells in nontransplant individuals with HCV.[41–43] Indeed, this association persists after liver transplantation and may be one of the paramount means for rapid disease progression in transplant recipients. Immunosuppressant therapy after transplantation likely leads to increased regulatory T cell expression that can contribute to the severity of recurrent disease.[44] Improved insight in immunosuppressive agents and the interaction between HCV and the immune system is needed to help tailor antiviral therapy and achieve successful treatment while minimizing adverse immunological effects.
Immunosupression is the cornerstone of patient and graft survival in liver transplantation.
However, intense immunosuppression promotes HCV recurrence and severity.[45–47] The treatment of acute cellular rejection with OKT3 or pulse-dosed corticosteroids lead to increased HCV RNA values, decreased graft survival and increased overall mortality. The concept that particular calcineurin inhibitors may impact the natural history of HCV in liver transplant patients is controversial.[48–50] Although overall survival is similar regardless of the calcineurin inhibitor use, preliminary data suggest that cyclosporine-based immunosuppressive regimens, when used in conjunction with interferon-alpha and ribavirin to treat HCV, may lead to a greater likelihood of virological response to therapy, possibly through greater T cell regulatory inhibition.[51] Further confirmatory studies are required.
Other factors that lead to worse outcomes in patients transplanted for HCV include prolonged cold and warm ischemia time (more then 90 min), recipient age, presence of diabetes, female gender and nonwhite race. Viral coinfection with human immunodeficiency virus has also been associated with more severe HCV recurrence and more rapid progression to cirrhosis.[52,53] HCV appears to be particularly more aggressive in recipients transplanted with higher MELD scores.[54] Emerging data suggest the recipient Interleukin-28b (IL-28b) genotype may also be associated with rapid histological recurrence,[55,56] although the exact mechanism is unclear. Interestingly, the IL-28b genotype does not appear to impact overall or liver-related survival.
Challenge of HCV Therapy and Minimization of Side Effects:
Early studies suggested that there was a potential for pre-emptive treatment as a method of preventing recurrent HCV disease and achieving a sustained viral response (SVR).[57] However, less than 20% of patients with genotype 1 who received pre-emptive treatment with a combination therapy including interferon resulted in a SVR. The current, standard of care regimen to treat recurrent HCV after transplantation consists of pegylated interferon plus ribavirin.[58] Few recipients are considered candidates for treatment early after transplantation. In one study, only 41% of 124 consecutive patients were candidates for empiric antiviral therapy within the first 6 weeks after transplantation.[59] The severity and frequency of neutropenia and anemia are much higher in transplant recipients. The risk of anemia in nontransplant patients is 15–20% and is approximately 40% in liver transplant recipients.[60] Tolerability is a significant limitation in recipients treated with preemptive antiviral therapy. Dose reduction and early discontinuation are common. Shergill et al. also demonstrated that 40% of the patients who received at least one dose of interferon discontinued treatment. Common reasons for drug discontinuation included acute rejection, pancytopenia, and depression unresponsive to medications. Ultimately, only 15% of patients achieved full-dose therapy and only 23% of patients took at least 80% of the treatment doses for at least 80% of the treatment duration. Challenges for the future are to limit side effects and increase the number of posttransplant patients with recurrent disease who are candidates for treatment. Lower SVR is achieved with pegylated interferon monotherapy; ribavirin monotherapy is ineffective in achieving a SVR.
Challenge of Achieving a SVR to Treatment:
A number of well-described factors have been shown to predict SVR in the nontransplant setting and several of these factors also appear to be predictive in the posttransplant setting. Like the pretransplant population, the most important predictor appears to be HCV genotype. Other factors include pretreatment HCV viral load, patient age, ability to maintain recommended target medication doses and duration of therapy. Although the polymorphism variant of the IL-28b gene region is related to severity of recurrent disease in preliminary studies, the polymorphism variant region is also associated with the likelihood of achieving a SVR.[61–63] This relationship has been well described in the nontransplant population, and early studies in transplant recipients with genotype 1 patients are promising.
The likelihood of achieving a SVR is strongly related to the ability to adhere to recommended doses. In the nontransplant setting, there is a substantial decrease in SVR when patients fall below a certain threshold of doses during treatment with pegylated interferon and ribavirin. Therefore, it is critical that patients and physicians adhere to the antiviral regimen, as long as it is safe to do so. Neutropenia and anemia are major limiting complications in liver transplant recipients. Consequently, only about 30% of treated patients are able to achieve target doses and duration of therapy.[58]
Although HCV is one of the most common indications for liver transplantation, patient survival is among the lowest of all indications for transplantation.[31] The ultimate goal of antiviral therapy in patients with recurrent HCV is to improve survival. Indeed, achieving a SVR can lead to fibrosis regression and improve survival. The regression of fibrosis can continue for several years after achieving a SVR.[64] However, the likelihood of achieving SVR among transplant recipients is between 25% and 45% (36,64–66). A 5-year survival of 92% can be expected among patients who are able to achieve a SVR compared to 66% in treatment nonresponders.[67] Infergen (interferon-alphacon-1) and ribavirin are approved for the treatment of interferon nonresponders in the nontransplant population. There is limited experience using this treatment combination in liver transplant recipients and further clinical studies are needed.
Challenge of Evaluating Protease Inhibitors as New Therapies for Recurrent HCV:
An incremental improvement in the treatment of HCV is the utility of protease inhibitors. In May 2011, two different protease inhibitors were approved by the Food and Drug Administration in the United States: boceprevir and telaprevir.[68,69] These protease inhibitors will be used in combination with pegylated interferon and ribavirin. In the nontransplant population, protease inhibitors lead to higher rates of SVR, and the duration of treatment may be shortened in patients who achieve a rapid viral response. Additional clinical studies are needed to define their role in the posttransplant setting. One limitation is the lack of data on drug interactions with antirejection medications used after transplantation. Protease inhibitors are associated with a high rate of anemia and this may be a significant limiting adverse effect as the risk of anemia with pegylated interferon and ribavirin already approaches 40% in liver transplant recipients.[60] The immunosuppressive medications used in the posttransplant population may exacerbate the bone marrow suppressive effects of antiviral therapy. Dose reduction of pegylated interferon and ribavirin may overcome this limitation but decrease the chance of maintaining an SVR. Growth factors such as granulocyte colony-stimulating factor and erythropoietin to treat neutropenia and anemia are promising alternatives.[70]
Challenge of Retransplantation:
Overall, retransplantation for HCV-associated graft failure is associated with a worse prognosis than primary transplantation for HCV. The role of retransplantation for patients who develop graft failure from recurrent HCV is controversial since retransplantation for recurrent HCV is associated with a worse prognosis than other indications for retransplantation.[71] Indeed, retransplantation for recurrent HCV disease may carry up to a 30% increased mortality risk. Factors associated with a favorable outcome after retransplantation include: MELD less then 21, portal HTN as sole manifestation of cirrhosis, short warm and cold ischemia times, previous SVR and retransplanted more then 30 days after initial liver transplantation.[72,73] Factors associated with worse outcome include: recipient age more them 55, renal failure, ventilator dependence, MELD more then 21, prolonged warm and cold ischemia times, early viral recurrence, and retransplantation between 8 and 30 days after the initial transplant. Retransplantation is a viable option for patients who have graft failure from recurrent HCV, and should be considered on a case by case basis.
Challenge of Removing Barriers to Liver Transplantation:
There are a number of barriers to transplantation in patients with chronic HCV and advanced liver disease. In Arizona, for instance, state funding for liver transplantation for patients with HCV cirrhosis was temporarily discontinued in October 2010. Even patients already listed were denied financial authorization for transplantation.[74] In California, retransplantation for cirrhosis from recurrent HCV is not covered by Medicaid. Moreover, the costs of antiviral therapy can be restrictive. Depending on the medication coverage, liver transplant recipients may not be able to afford pegylated interferon and ribavirin even though these antiviral therapies have been found to be cost-effective in the posttransplant setting.
Proper Selection of Transplant Recipients With HCC
HCC is the fifth most common cancer worldwide. It develops in the context of cirrhosis in 80% of patients. Current recommendations are to enroll patients who are at risk for HCC in a surveillance program. Screening tests fall into two categories, serological and radiological. As a screening test, ultrasonography is the most commonly used modality and has been reported to have sensitivity between 65% and 80% and specificity greater than 90%. Alpha-fetoprotein (AFP) is the most studied serological marker for HCC. A serum AFP level of 20 ng/mL provides a sensitivity of 60%, which is suboptimal for general use as a screening test alone. AFP still has a role in the diagnosis of HCC, and one challenge for the future is to discover new biomarkers with appropriate predictive value to better screen for and diagnose HCC.
Challenge of Developing New Biomarkers for HCC:
Diagnostic serological markers, prothrombin induced by Vitamin K Absence II (PIVKAII) and the ratio of glycosylated AFP L3 fraction (AFP-L3) to total AFP, have been in existence for nearly 20 years. The former has been shown to inversely correlate with prognosis, as well as portal vein invasion, tumor size, intrahepatic metastasis, tumor recurrence and patient survival after treatment. AFP-L3 has also been shown to be useful in evaluating tumor recurrence and malignant potential as well as prognosis. Glypican-3 is a heparin sulfate proteoglycan bound to the cell surface and is highly expressed in fetal, but not normal, adult liver. Expression of Glypican-3 has been observed in HCC tumors that fail to express AFP and therefore may be useful in evaluating liver masses in the context of a normal serum AFP. However, in one report, serum Glypican-3 had a sensitivity of 46.7% and a specificity of 93.5%.[75] A goal for the future is to develop improved biomarkers for HCC screening and diagnosis, which may allow for earlier detection.
Challenge of Developing Improved Risk Stratification Models for HCC:
Only 20% of patients with HCC in the United States are candidates for surgery (resection or transplantation) at the time of diagnosis. Resection and liver transplantation are the best, curative options to treat HCC with liver transplantation having the added benefit of removing the underlying cirrhosis that provides an optimal environment for hepatocarcinogenesis. Liver transplantation for HCC in the late 1980s and early 1990s achieved poor results with 5-year survival ranging from 20.4% to 35.6% and was attributed to selection of recipients with advanced stage cancer.[76,77] In 1996, Mazzaferro and colleagues proposed the Milan Criteria (one tumor less than 5 cm or no more than three tumors with each no more than 3 cm) that translated to a patient survival rate of 75% at 4 years.[78] The MELD scoring system began in 2002, and exception points were granted for HCC tumors that fell within the Milan Criteria. The MELD exception led to an increase in the proportion of patients transplanted for HCC from 20% in 2002 to 27% in 2008. The promising results for liver transplantation in early-stage HCC led to a study by Yao and colleagues at the University of California, San Francisco, concluding that a modest expansion of the criteria to one tumor up to 6.5 cm or no more than three tumors, none greater than 4.5 cm with a total diameter of less than 8 cm, could maintain survival while loosening the criteria for transplantation.[79] A separate retrospective study of 467 patients from the University of California, Los Angeles, showed similar survival between the Milan Criteria and the UCSF Criteria, arguing that exception points should be expanded to patients meeting the less restrictive standard.[80]
There are several prognostic models for HCC that all rely on a combination of radiographic appearance (number of tumors and size), histological assessment (vascular invasion) and overall liver function. Personalized genomic profiling of liver tumors may provide an individualized molecular signature to guide treatment. Such a molecular pattern would link tumor biology to therapy. Studies looking at selected molecular markers of dysregulated signaling pathways and matching those defects to specific inhibitor therapies are already underway in a limited capacity.[81]
The challenge for liver transplantation in HCC is to define an improved preintervention staging algorithm based upon morphologic and genetic criteria that is linked to a personalized cancer treatment program ranging from surgery to the use of new, systemic molecular inhibitors to fight cancer. A new evaluative criterion would include size and number of lesions in addition to expression or mutation of critical genes leading to increased cell cycle progression and metastatic potential.
Several investigators have used microarray technology to sample HCC tumors, dysplastic tissue, or normal surrounding tissue to understand the genetic variation as cells progress toward carcinoma in the liver. The variability in disease progression among patients with HCC may be due to genetic heterogeneity across the population at large. Genetic expression patterns among individual cancers may be able to reduce this complexity by distinguishing between different subgroups of HCC, allowing tumors to be stratified into low and high aggressive potential that conventional histopathology may be unable to accomplish. One such study by Lee and colleagues analyzed gene expression patterns in HCC from rodents and humans and found that the expression of certain genes in aggressive HCC tumors were shared with fetal hepatoblasts, suggesting that an immature cell of origin may confer poorer prognostic consequences.[82]
Vascular invasion is an unfavorable prognostic factor in HCC.
Occasionally, macrovascular invasion can be noted on preoperative imaging. However, microvascular invasion can only be detected upon histopathological analysis. The presence of vascular invasion is an exclusion for MELD exception points in patients waiting for orthotopic liver transplantation. The detection of gene expression indicating a high likelihood of vascular invasion would be an effective preoperative assessment and screening tool if it can be reliably used as a surrogate for histopathology and survival posttransplant. Ho and colleagues evaluated gene expression in 18 patients undergoing curative resection for HCC. Fourteen discriminative genes were found and could accurately predict patients with and without vascular invasion in a test group. Among the genes that were identified, three were related to cell growth and others to oxidative DNA damage.[83]
Biomarkers have yet to be incorporated into a formal risk stratification scoring system. However, Kaseb and colleagues have investigated the use of serum vascular endothelial growth factor (VEGF) and applied it to the Cancer of the Liver Italian Program (CLIP) score. The CLIP staging system is based upon liver function (Child's Score) and tumor characteristics including tumor morphology, extension, AFP level and portal vein thrombosis. Although this study was conducted in patients with mostly advanced, unresectable cancer, the authors demonstrated that higher levels of the VEGF (more then 450 ng/mL) led to poorer survival in each of four CLIP score levels. Differences were significant at advanced levels of disease (score 3–4) but poorer survival trended toward those with elevated VEGF levels throughout.[84] These modified scoring systems, based upon tumor biology, require further validation before they are applied to patients. However, a more immediate effect may be seen in clinical trials where the ability to further distinguish patients and predict prognosis based upon the individual biology of their tumor may demonstrate a subset of patients that benefit from a given therapy.
Challenge of Developing Novel Molecular Inhibitors to Treat HCC:
There is a new emphasis on understanding the complex signaling pathways in HCC in hopes of identifying molecular inhibitors to halt disease progression. Sorafenib is the prototypical multikinase inhibitor that targets multiple signaling pathways including: Raf serine/theonine kinase and the VEGF Receptor 1-3, platelet-derived growth factor receptor-B (PDGFR-B), c-kit, FMS-like tyrosine kinase 3 (FLT-3) and p38 tyrosine kinase. Sorafenib is able to halt growth and induce apoptosis in HCC cell lines. Two phase III clinical trials have shown a survival advantage in patients receiving Sorafenib in North America, Europe and Asia.[85,86] The SHARP study analyzing North American and European patients showed that median survival is extended from 7.9 to 10.7 months (hazard ratio 0.69, p less then 0.001) with Sorafenib therapy.[85] What is needed is a multicenter trial analyzing Sorafenib as adjuvant therapy in HCC and liver transplantation.
Given the modest benefit in survival with Sorafenib and the discovery of multiple molecular pathways that give rise to HCC, novel molecular inhibitors are in the drug development pipeline and several clinical trials to evaluate combination therapies are ongoing such as Sorafenib, Gemcitabime (NCT 00844688) and Sorafenib and trans-arterial chemoembolization (NCT 00618384). New potential targets for inhibition include growth factor receptors and signal transduction pathways that govern proliferation, motility and the metastatic potential of tumor cells. Candidate downstream targets include Raf, MEK and extracellular signal-regulated kinase of the Ras pathway, the phosphatidylinositol 3-kinase (PI3K)–AKT–mammalian target of rapamycin (mTOR) pathway and the Janus kinases/Signal transducers and activators of transcription pathway.
A primary consideration for new multikinase inhibitors is to select those with an appropriate balance between anticancer efficacy and side effects that may be related to the broad activity of these agents. Sunitinib is an oral multikinase inhibitor of receptor tyrosine kinases VEGF Receptor 1–3, PDGFR-a, PDGFR-B, FLT-3, c-kit, RET and CSF receptor type 1. Sunitinib suppressed tumor growth of five HCC xenografts[87] and demonstrated a progression-free survival of 3.9 months and an overall survival of 9.8 months in a phase II trial.[88] However, higher doses of Sunitinib have been associated with an increase in hematological complications.[89] A phase III trial to evaluate Sunitinib versus Sorafenib in patients with advanced HCC using a modified Sunitinib dosing regimen was terminated in April 2010 due to an increased incidence of adverse effects with Sunitinib and failure to demonstrate comparable efficacy to Sorafenib (http://www.clinicaltrials.gov, NCT 00699374). The early discontinuation of this trial is certainly a set-back for the introduction of new tyrosine kinase inhibitors for clinical use against HCC.
The PI3K/AKT/mTOR pathway is a major protein cascade involved in immune regulation and carcinogenesis. Activation of this pathway indicates poor prognosis in patients with HCC. Mutations in the catalytic domain of PI3K lead to enhanced AKT expression and activation of mTOR. Rapamycin, an immunosuppressant, inhibits replication of HCC cell lines in vitro and expansion of HCC tumors in animal models. A study analyzing rapamycin in postliver transplant patients transplanted for HCC showed improved survival at 6 years compared to counterparts not receiving this drug, suggesting that rapamycin may have both anti-inflammatory and anti-tumor properties.[90] A new rapamycin analog, Everolimus (RAD001), is an oral medication for anti-HCC therapy and has been used in the posttransplant setting in patients at high risk for HCC recurrence, calcineurin toxicity and refractory rejection.[91] Several clinical trials valuating the efficacy of Everolimus in HCC are ongoing (Table 2).
The deregulated expression of membrane bound proteins in HCC may serve as a ligand for targeted drug therapy. Lee and colleagues at the University of Hong Kong showed that HCC cells with increased expression of the epidermal growth factor receptor can be specifically targeted for gene therapy to replace mutated p53.[92] Other groups have used bionanocapsules consisting of hepatitis B virus surface antigen molecules within a lipid bilayer to specifically deliver DNA to liver tissues.[93] Targeted drug delivery systems are important areas of development that concentrate a therapeutic agent in tumor cells while minimizing exposure to healthy tissues.
Concluding Remarks
The success of liver transplantation has presented the field with several challenges, namely the need to overcome an inadequate supply of useable donor organs and the importance of proper recipient selection. Solutions to expand the donor pool will require the use of more ECD and DCD organs. Additional studies are needed to further evaluate optimal selection criteria, including the use of steatotic grafts, and proper donor to recipient matching.
Recipient selection is especially important for patients with HCV and/or HCC. For those with HCV, better medications with favorable side-effect profiles are needed to establish a SVR and increase survival in posttransplant patients with recurrent disease. Hopefully, protease inhibitors will be tolerated in the posttransplant setting, but anemia remains a concern.
For HCC, earlier, improved detection may translate into better survival since early disease is best treated by a surgical approach. The new paradigm of targeting molecular inhibitors to the critical switches that turn on liver cancer is being aggressively pursued for those with advanced disease. This multitargeted strategy will utilize predictive molecular biomarkers and combinations of molecular signaling inhibitors to block the deregulated cell cycle machinery of cancer cells. Taken together, the outlook for liver transplantation remains promising. History has shown that the field has met and surpassed clinical challenges that make liver transplantation the success and proven life-saving treatment for patients that it is today. We have identified future challenges that, if met, will further expand transplantation and increase survival among liver recipients.




No comments:
Post a Comment